Lots on vaccines and treatments. Doubter docs and illustrious superspreaders. Mysteries of New York mayors, Delta Airlines, and Italy. A rare Fauci slip-up... plus the inevitable extras.
Treatment news
 |
| Fetching a box of Paxlovid |
Paxlovid: Even in the United States, where there’s plenty available, most Paxlovid doses are remaining on pharmacy shelves even while those most in need are often unable to access it. Unless, of course, they’re wealthy and well-connected – take Kamala Harris. She was given a course after testing positive for SARS-CoV-2, despite being young, healthy, and asymptomatic.
On the positive side, use is picking up now, with prescription rates in the US more than doubling month by month and more than 500,000 antiviral courses used by April 25th. By now the drug has been taken by about 0.6% of all patients with confirmed cases.
Italy is no longer making potential users go to a hospital to consult an infectious disease specialist and pick up the box of pills, but instead trusts primary care physicians to prescribe it correctly and have ordinary pharmacies stock it. This may or not be a wise choice given the limited training of Italian GPs. As of a month ago only 8300 Italians had received Paxlovid, 0.15% of confirmed cases. So we can roughly estimate that if the drug is being underused in the USA, the underuse is 4 times worse in Italy.
On the negative side: To start with, when given to healthy people living in the same household with COVID-19 cases, Paxlovid doesn’t keep them from getting sick – I don’t see why anybody would have thought it could. Also, if the drug is used very widely it could push the SARS-CoV-2 virus to evolve into drug-resistant strains, as happens with antibiotics. Furthermore, the studies showing brilliant results for antivirals were performed in unvaccinated COVID-19-naïve patients, so we really don’t know how effective these drugs are in preventing patients who have some immunity from vaccination or previous infections, who by now are most people in the Western world – as one commentator wrote, “Show Us The Data!”
Finally, about 2% of patients have some degree of relapse after completing a course of Paxlovid. Most of the time that just means antigen tests returning positive, but in other cases the person actually becomes ill again. What to do? Ignore the relapses, given that they are almost never severe and that the patient’s own immune system is likely to kick in and knock out the virus? Prolong the standard treatment from 5 days to 6 or 7? Or offer symptomatic relapsers a second 5-day course?
Convalescent plasma: If you’re confused about whether infusions of convalescent plasma help COVID-19 patients, you’re not alone. Two experts think we still don’t know, and try to make sense of various discrepant results in a New England Journal of Medicine editorial. Probably locally-sourced plasma could decrease deterioration in high-risk patients by 25%-50%, and could find appropriate use in developing countries lacking easy access to antivirals.
Remdesivir: The final results from the World Health Organization’s large Solidarity trial clarify its role in hospitalized patients: useless for patients already on ventilators, but capable of slightly decreasing the risk of winding up on one. This borderline effect is far inferior to its splendid performance in outpatients.
Inhaled interferon beta: After good results in a Phase 2 trial among hospitalized patients, Synairgen’s innovative product has cratered in Phase 3, not getting patients discharged faster or preventing mechanical ventilation or death. Some subgroups of patients did seem to benefit, so the company hasn’t yet thrown in the sponge.
S-217622: A new oral antiviral developed in Japan hastens recovery from COVID-19 . . . if you’re a hamster. In a small Phase 1-2 trial in human patients, it helped clear the virus more quickly but did little or nothing in the way of symptom relief. A multinational Phase 3 trial could conceivably come up with better results.
Ensovibep: This Swiss intravenous antiviral is new to me, though Novartis announced promising Phase 2 results (78% reduction in clinical worsening) back in January. It had already flunked out in hospitalized patients, and the company says it may not be willing to go forward with the Phase 3 trials that would be needed for authorization in the US or the EU.
Vaccine news
 |
| Peter Hotez and Maria Elena Bottazzi, the developers of Corbevax |
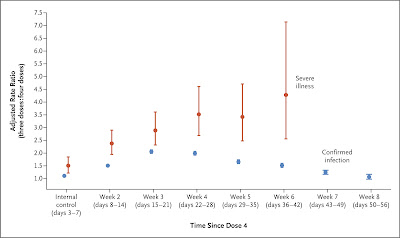
Second boosters: The World Health Organization is now saying they are probably beneficial for “highest risk groups,” especially the immunosuppressed, while emphasizing how little we know. The CDC, on the other hand, is expanding its recommendations, advising everybody 50 and over to get a second booster 4 months after their first one. We now have some important new data on real-world effectiveness from 3 Israeli studies performed during the Omicron surge.
 |
| Israeli study #1 |
The first looked at all 1,252,331 citizens over 60 who had received a first Pfizer booster dose 4 or more months earlier and were eligible for a second one. Those who got it were half as likely to become infected with SARS-CoV-2 than those who had not, but the benefit wore off by 7 weeks. They were also a quarter as likely to develop severe COVID-19, at least for 6 weeks. This was not a randomized study, though, and people who chose to get their 4th shot were older, and probably sicker, than those who didn’t.
 |
| Israeli study #2 |
The second, from a large insurance company, had a relatively long 10-week follow-up on 97,499 people over 60, of whom about 1/3 received a second booster. The 4th dose gave an additional 64% protection against infection, which began to wane a month after the shot and by 10 weeks had fallen to 29%. Protection against severe COVID-19 and death, however, if anything strengthened over the course of the study, rising from 73% at one week to 86% at 7 weeks. This study did have health information, and confirmed my suspicion that individuals who chose to receive a second booster had more chronic conditions than those who did not.
 |
| Israeli study #3 |
The third study, from a different insurance company, had similar findings, though only over 40 days, on 563,465 older people, of whom 58% had received that second booster. The 4th dose lowered hospitalizations by 64% and deaths by 78% as compared with 3 doses.
Having had a 4th shot myself last month I find these studies somewhat reassuring, in that the 10-week boost in preventing severe disease is less fleeting than I’d feared. But the second booster seems to wane sooner than the first one, the first one waned sooner than the original course of vaccines, and I fear this pattern will continue with future boosters of the same mRNA vaccines. We can’t be going back for boosters every month or two. So I still think that until we have boosters that give multivariant immunity individuals who are neither elderly, immunocompromised, or chronically ill should hold off on those second boosters unless they’re about to travel, attend a conference with unmasked people, or otherwise be put at particularly high risk.
Johnson & Johnson: The news is mixed. Good: two doses were just as effective as 2 doses of Pfizer against severe disease during the Omicron surge in South Africa. J&J should probably have been two-dose all along. Bad: the US Food and Drug Administration has further limited its use, citing the risk of thrombosis with thrombocytopenia syndrome (blood clots), which have been documented in 60 Americans and killed 9. Now the FDA recommends it be given only to adults who are ineligible for mRNA vaccines, cannot obtain them, or say J&J is the only vaccine they would accept.
Valneva: The makers of this French whole-virus vaccine already got my goat by palming off Phase 1-2 trial results as “Phase 3.” There is still no true Phase 3 trial in the works and the European Union has cancelled its contract, though the United Arab Emirates and perhaps the UK will give it a try.
Convidecia (CanSino): The World Health Organization has green lighted emergency use of this one-dose Chinese viral vector vaccine on the basis of Phase 3 trials showing 58% efficacy against symptomatic wild-strain COVID-19; unfortunately only 7.9% of trial volunteers were over 60. It’s the 11th COVID-19 vaccine to get the WHO nod.
Novavax: Unbeknownst to me, this vaccine arrived in Europe, including Italy, toward the end of February. It hasn’t exactly caught on, rarely hitting 300 doses a day in Italy (vs. Pfizer’s 30,000+). It may soon get an Emergency Use Authorization in the United States as well.
Covifenz: The new Canadian vaccine I mentioned a few posts back, touted as “plant-based” because it uses tobacco plants to produce the spike protein, has now been confirmed in a Phase 3 study to be 70% effective against symptomatic COVID-19 from variants up to and including Delta, with no vaccine recipients developing severe disease. Virtually all the subjects were healthy adults under 65, so we know nothing about this vaccine’s performance against Omicron, in older people, or in the chronically ill. It’s already been approved for use in its home country, but I’m not sure anybody else will bite.
ZF2001: Still another new entry, from China, is a protein subunit vaccine like Novavax. In Phase 3 trials it’s 76% protective against COVID-19 for 6 months, and more than 85% against severe outcomes and death. Like Covifenz, it’s only been tested in working-age populations and not at all against Omicron.
Corbevax: This is the open-source, inexpensive, vegan, halal Texas-made protein subunit vaccine. In India it’s been approved in India even for kids – though not without backlash – and also in some African countries, despite the lack of any Phase 3 trial data.
Too much of a good thing? That makes 4 new COVID-19 vaccines hitting markets within months. Does the world really need them? Two New England Journal of Medicine editorialists argue that the answer is yes, largely because mRNA vaccines are complicated to store and deliver, whereas these new ones are both convenient and cheap.
Dream on: Eric Topol, one of the most intelligent commentators around, has written a Guardian piece calling for something very simple: variant-proof nasal vaccines. But the simplicity is only apparent, because all attempts thus far to develop any vaccine that’s even slightly variant-proof have been dead in the water.
Kids: COVID-19 vaccines are relatively ineffective in children, both the ones from China and the hyper-low-dose one from Pfizer whose benefit is also particularly short-lived. Moderna has now performed a Phase 3 trial of a half-dose vaccine – identical to the adult booster dose – in about 3000 children between 6 and 11, and early results are very promising: efficacy of 73% against SARS-CoV-2 infection (Delta variant) and about 90% against symptomatic COVID-19, without substantial side effects. Protection began 2 weeks after the first dose and showed no sign of waning over 3 months of follow-up; since the Moderna product contains 50 mcg of mRNA vs. Pfizer’s 10, we can guess it will be longer-lasting.
Variant watch
Transmissibility: An interesting review and meta-analysis shows that after a very rapid rise early in the pandemic when we knew little about COVID-19, the rate of transmission from patients to family members has continued to climb, especially after the more infectious Delta and Omicron variants hit town.
Omicron subvariants BA.2.12 and BA.2.12.1: The latest twists on the Omicron variant are coming ‘round themountain. They are even more transmissible than BA.2, and ever more capable of infecting people who’ve been triple- or perhaps even quadruple-vaccinated, and who have recovered from previous bouts of COVID-19. The latest variants can even attack people who’ve had previous versions of Omicron. BA.2.12.1 now accounts for 1 in 3 new cases in the United States, with virtually all the rest BA.2, and for two-thirds of cases in New York City. Then there’s the even newer BA.4 and BA.5 subvariants – they are already dominant in South Africa and present in more than a dozen other countries. Remember the optimistic predictions early on that SARS-CoV-2 wouldn’t mutate as fast as, say, the flu virus, so vaccines would last for years? If only!
Omicron XE: Another hypertransmissible variant, combining bits of the genetic material of both BA.1 and BA.2. There are handfuls of cases in the UK and Japan. But other chimera variants such as Deltacron never got anywhere, so I suspect this one won’t either.
A Fauci ouchie
Anthony Fauci told an interviewer on the PBS Newshour on April 26th,“We are certainly right now in this country out of the pandemic phase” of COVID-19. The very next day he had to backtrack, telling NPR, “I probably should have said 'the acute component of the pandemic phase.'” And telling the Washington Post: “out of the full-blown explosive pandemic phase” and into “a transitional phase, from a deceleration of the numbers into hopefully a more controlled phase and endemicity.”
My own spectacles are less rose-tinged than Fauci’s. For one thing, the running average of COVID-19 deaths in the USA, while on its way down, is currently 339 per day. On a yearly basis that would be more than quadruple the death rate from influenza, often considered an appropriate criterion for when COVID-19 can be considered a “normal” and merely endemic, infectious disease. This is far from “controlled.”
Plus the Omicron subvariants are getting more and more contagious, and as long as the developing world remains unvaccinated – only 17% of Africans are “fully vaccinated” – new and possibly more virulent variants are sure to pop up.
Superspreading
Nancy Pelosi picked up the SARS-CoV-2 virus at this White House event celebrating the Affordable Care Act; she says she never felt sick. Obama had had a mildly symptomatic case 3 weeks earlier.
More than 70 people, from Merrick Garland, Susan Collins, and Eric Adams on down, tested positive for COVID-19 days after attending this year’s white-tie Gridiron Club dinner.
I caught my husband (in the purple cap) and almost everybody else with their masks down in front of the “A Mask Is Still Required” sign at the airport in St. Thomas.
Even smart people are sometimes dumb.
Doubter docs
 |
| Dr. Bruce Boros, who fell seriously ill with COVID-19 after a novax medical conference |
A shocking poll of American primary care physicians found that 10.1% didn’t agree that COVID-19 vaccines are safe and 9.3% didn’t agree they are effective. That’s fewer doubters than in the US population as a whole, but still it’s pretty scary, especially since PCPs are supposed to be the backbone of the US vaccination campaign. Unsurprisingly, docs who identify as politically conservative were more likely to be skeptics. But so were Hispanic docs – go figure.
Delta Airlines interprets the CDC
Checking in for a flight, here’s what Delta forced us to “Acknowledge”:
THE STANDARD FOR SAFER TRAVEL
DELTA'S COMMITMENT TO YOU
The Delta CareStandard℠ focuses on creating a safer experience for everyone.
YOUR COMMITMENT TO SAFETY
If traveling within the United States, I/we confirm that I/we are not currently required to isolate or quarantine due to COVID-19 exposure, symptoms or a positive diagnosis under the current CDC COVID-19 guidelines.
If traveling internationally, I/we confirm that I/we are not required to isolate or quarantine due to COVID-19 exposure, symptoms or a positive diagnosis under the current public health regulations at the origin and destination.
Here are the actual current CDC Guidelines:
Then there’s the “trip details” sent 3 days before our flight from St. Thomas, which included
In fact, as per the huge sign in the St. Thomas airport and Gov. Hochul’s continuing masking requirement in all New York State transportation hubs, all that’s true is the “On Board” part.
Fire that fact checker!
COVID eats the Big Apple
 |
| New York Mayor Eric Adams vowing not to reinstate mask mandates |
With 6 times as many cases being officially reported as in March and 3 times as many hospitalized, New York City has already entered the Orange or high COVID risk zone, even though cases are vastly underestimated because few home tests get reported. Under the older, better, CDC criteria, New York would have been “high-risk” months ago. Theaters are again having to cancel performances. Mayor Adams’ reaction? Mask mandates in preschools, of all things, and some vaccination checks. He and his health commissioner stubbornly refuse to take the really vital step of mandating masking in all public indoor settings, even though that mandate is official New York City policy and health experts are asking “How does this make any sense, @NYCMayor?” and “Wow, when are we going to learn.” At least Adams can’t contravene Governor Hochul’s statewide mandate for masking on public transport, which – eyeballing it in late April – is being obeyed by about 90% of subway riders.
On a personal note: I attended an art opening on April 30th at the Alexandre Gallery downtown. A door sign says masks are obligatory, but none of the staff and almost nobody else were wearing them. The artist, the late Edith Schloss, was an intimate friend and I had been looking forward enormously to the show, but it felt so unsafe that I walked out after 5 minutes. I was unsurprised to hear a few days later that multiple attendees had come down with COVID-19.
The rest of the country isn’t doing any better. Even according to those new CDC guidelines that vastly underestimate how much virus is around, 45% of Americans are now in counties rated medium- or high-risk, so should probably be wearing masks indoors, up from 32% just 4 days ago and 1% in mid-March. If the older, better guidelines were still in use 73% of counties would be counted as high-risk rather than just 24%. And the CDC is predicting both hospitalizations and deaths will keep rising at least through the middle of June, even though Omicron is usually mild. (They’ve now officially given up on trying to forecast case numbers.)
The Italian mystery
In Italy more than 90% of everyone over 12 is fully vaccinated, everybody masks indoors, and Green Passes were essential until May 1st, and yet the country has had higher total per capita death rates than any other Western country except Belgium (neck-and-neck) and the USA (alas). One reason is surely the horrific early epidemic in Northern Italy, which overwhelmed hospitals at a time when virtually nothing was known about the disease, including how it was transmitted.
But the death rate is still worse than other major EU countries’ and is now running double the rate in the US. In large part this may be because Italy is barely making any use of antiviral drugs such as Paxlovid and remdesivir. But I fear it’s partly due to medical incompetence – as per a recent article in the Sole 24 Ore newspaper. I have long considered the care in many Italian Emergency Rooms and intensive care units to be subpar. Some try to put the blame on Italy’s relatively elderly population, but Germany has seen 43% fewer deaths despite a similar age distribution.
When do breakthrough infections go sour?
COVID-19 in fully vaccinated people is usually mild, but not always. Who does well, and who gets very sick or dies? A Morbidity and Mortality Weekly Report examined that question among 2246 fully vaccinated individuals with breakthrough infections, and identified 8 risk factors: age over 65, immunosuppression, diabetes, and lung, liver, kidney, neurological, or heart disease. Every patient with a severe case had at least one of those risk factors, and 78% of those who died had 4 or more. Sex, race, and time since vaccination had no impact. Being a COVID-19 survivor, monoclonal antibody treatment, and booster shots were all protective factors, and Moderna worked better than Pfizer or J&J. But never forget, the most important risk factor for getting severely ill or dying from COVID-19 is being unvaccinated.
My new favorite conspiracy theory
Nobel laureate Luc Montagnier, who died in February, said lots of nutty things about lots of subjects including COVID-19 (the virus was created in a laboratory, it incorporated genetic material from HIV, etc.) but there is no evidence that this is one of them. The claim is of course B.S.: vaccine boosters can't give you AIDS.














No comments:
Post a Comment